At Cambridge Bioscience, we offer a range of fecal collection kits for the convenient collection of fecal material for downstream nucleic acid analysis. This includes DNA/RNA Shield, a nucleic acid preservation reagent as well as the Bunny wipe sample collector and ColOff which provide user friendly methods for at home collection.
The DNA/RNA Shield Fecal Collection Tube, powered by the DNA/RNA Shield sample stabilisation reagent captures a microbial snapshot of each sample while effectively deactivating viruses, ensuring samples are safe and primed for transportation.
The Zymo Research DNA/RNA Shield™ Fecal Collection Tubes (DX) are certified for in vitro diagnostic applications, meeting CE-IVD standards. These tubes come pre-filled with DNA/RNA Shield reagent, facilitating ambient temperature shipment while preserving DNA, RNA, and deactivating pathogens in fecal samples. Samples stored in these tubes remain stable at room temperature and can be frozen for extended storage . Zymo Research also offers customizable solutions for customers seeking tailored collection kits for specific applications.
The DNA/RNA shield fecal collection tube has become the #1 most cited fecal collection technology with over 5000 peer-reviewed citations in scientific publications, including Nature.
Benefits of using the DNA/RNA Shield Fecal Collection Tubes (DX)
- CE-IVD certified

- No reagent removal required prior to nucleic acid purification
- Pre-filled with DNA/RNA Shield
- RNA stable at room temperature for one month
- DNA stable at room temperature for two years
- Scoop attached to screwcap of tube
- Available with/without nylon swab
- Customisable solutions available
Bunny Wipe™ Fecal Sample Collector
 Zymo’s Bunny Wipe Fecal Sample Collector is designed as a user friendly, toilet paper like device that simplifies at home fecal sample collection for downstream nucleic acid analysis. After wiping, the lower strip of the wipe is peeled off and placed into a DNA/RNA Shield fecal collection tube, preserving DNA and RNA at ambient temperatures for downstream processing such as for NGS, qPCR or microbiome studies.
Zymo’s Bunny Wipe Fecal Sample Collector is designed as a user friendly, toilet paper like device that simplifies at home fecal sample collection for downstream nucleic acid analysis. After wiping, the lower strip of the wipe is peeled off and placed into a DNA/RNA Shield fecal collection tube, preserving DNA and RNA at ambient temperatures for downstream processing such as for NGS, qPCR or microbiome studies.
Key features:
- Familiar wipe format – functions like regular toilet paper for intuitive and sterile sample collection
- Flushable – the water soluble wipe can be flushed with standard toilet paper, eliminating additional waste
- Peeled collection strip – user removes a designated strip containing the fecal material for accurate sampling
Microbial Composition of Stool is Unchanged After One Month at Ambient Temperature with DNA/RNA Shield™
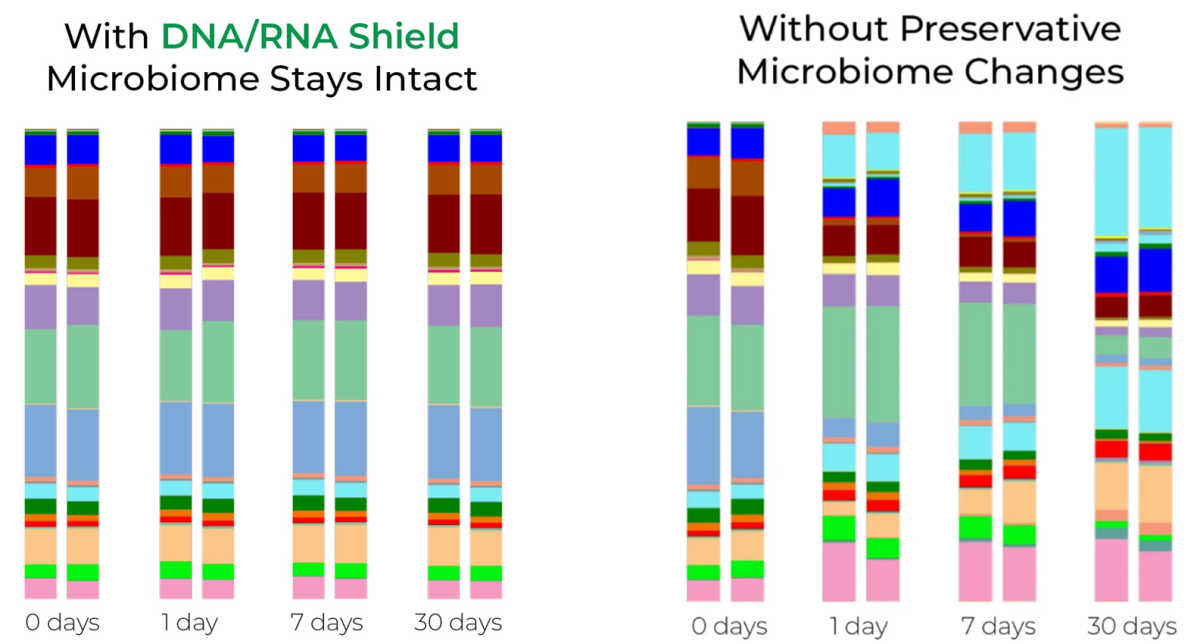
Stool samples suspended in DNA/RNA Shield™ and stored at room temperature were compared to stool without reservative for one month. They were sampled at the indicated time points and processed with ZymoBIOMICS® DNA Mini Kit. The extracted DNA was then subjected to microbial composition profiling via 16S rRNA gene targeted sequencing. Samples stored with DNA/RNA Shield™ had a constant microbial composition while the samples stored without shifted dramatically.
| Device specifications | 20 x 76 mm screw cap tube prefilled with DNA/RNA Shield (9 ml). Spoon attached to screwcap. |
|---|---|
| Device storage | RNA: Ambient temperature (4°C-25°C) > 1 month DNA: Ambient temperature (4°C-25°C) > 2 years DNA & RNA: Frozen (< -20°C): Indefinitely |
| Registration status | CE-IVD certified (R1101-E) |
| Sample collection | 1g or 1mL stool sample |
| Sample source | Stool, soil, environmental samples |
The DNA/RNA Shield™ Fecal Collection Tubes are also available in a Research Use Only format.
DNA/RNA Shield maintains the microbial composition after multiple freeze-thaw cycles
High-quality DNA from stool stored in DNA/RNA Shield after up to 10 freeze-thaw cycles. Microbial compostition profiiing via 16S rRNA gene targeted sequencing.
DNA/RNA Shield: complete pathogen inactivation
DNA/RNA Shield has been rigorously tested to ensure its capability to inactive even the toughest of viruses. In an independent study, the virucidal activity was shown to inactivate murine parvovirus1. DNA/RNA Shield abides by the Center for Disease Control’s (CDC) guidelines for pathogen inactivation2.
DNA/RNA Shield effectively inactivates pathogens. This includes tough-to-lyse microbes or viruses without the need for additional steps, such as heat-treatment, homogenisation, or alcohol sterilisation. DNA/RNA Shield has been rigorously tested to ensure inactivation of pathogens including:
Bacteria
B. subtilis, E. faecalis, E. coli, L. fermentum, L. monocytogenes, M. tuberculosis, P. aeruginosa, S. enterica, S. aureus, S. pneumoniae, X. fastidiosa
Viruses
Parvovirus, Chikungunya virus , Dengue virus, Ebola virus, Herpes simplex virus-1, Herpes simplex virus-2, Influenza A, Rhinovirus, MERS-coronavirus, West Nile virus
Yeast & eukaryotes
C. albicans , C. neoformans , S. cerevisiae , P. malariae

1. Dr. Thraenhart and Dr. Jursch. Virucidal activity of the nucleic acid preservation product “DNA/RNA Shield™” against the murine parvovirus (MVM) at 20 °C.
2. Guidance on the inactivation or removal of select agents and toxins for future use. Center for Disease Control (CDC)
Material available to download
DNA/RNA Shield brochure
DNA RNA Shield Fecal Collection Tube (DX) instructions for use
DNA/RNA Shield Fecal Collection Tube (RUO) instructions for use
DNA/RNA Shield citations
Products
Note: product availability depends on country - see product detail page.
| Details | Cat number & supplier | Size | Price |
| DNA/RNA Shield Fecal Collection Tube – Dx (10 pack) CE-IVD R1101-E · Zymo Research | R1101-E Zymo Research |
10 pce |
£138.00
10 pce
view
|
| DNA/RNA Shield Fecal Collection Tube (10 pack) R1101 · Zymo Research | R1101 Zymo Research |
10 pce |
£138.00
10 pce
view
|
| Bunny Wipe with DNA/RNA Shield Fecal Collection Tube (10 pack) R1138 · Zymo Research | R1138 Zymo Research |
10 pce |
£174.00
10 pce
view
|
| Bunny Wipe Fecal Sample Collector (1 wipe) R1133-1 · Zymo Research | R1133-1 Zymo Research |
1 pce |
£9.00
1 pce
view
|
| DNA/RNA Shield Fecal Collection Tube (with Beads) R1137 · Zymo Research | R1137 Zymo Research |
10 pce |
£138.00
10 pce
view
|
| Feces Catcher (10 pack) R1101-1-10 · Zymo Research | R1101-1-10 Zymo Research |
10 pce |
£14.00
10 pce
view
|
| DNA/RNA Shield Fecal Collection Kit R1180 · Zymo Research | R1180 Zymo Research |
1 pce |
£21.00
1 pce
view
|
| ColOff (Stool Collection Device) (5 pack) R1101-2-5 · Zymo Research | R1101-2-5 Zymo Research |
5 pce |
£40.00
5 pce
view
|
| Bunny Wipe Fecal Sample Collector (10 wipes) R1133-10 · Zymo Research | R1133-10 Zymo Research |
10 pce |
£75.00
10 pce
view
|


