- In this section:
- Contact our blood specialists
- Blood service testimonials
 Fresh or frozen, high-yield leukocyte fractions for dependable research
Fresh or frozen, high-yield leukocyte fractions for dependable research
Fresh buffy coats collected from healthy, rigorously screened donors and processed promptly to ensure high cell yield and reliable performance. Each unit includes full donor metadata and is delivered with flexible logistics to support time-sensitive studies across the UK.
 Why choose Research Donors human buffy coat?
Why choose Research Donors human buffy coat?
- High-yield leukocyte fraction – rich in lymphocytes, monocytes, and granulocytes, supporting a broad range of immune and genomic workflows.
- Well-characterised, fully screened donors – medication-free donors with CMV/EBV testing, optional HLA typing, and clear demographic data to ensure study consistency.
- Consistent quality – collected and processed using validated protocols to ensure reproducible cell composition and reliable downstream performance.
- Dependable logistics and turnaround – next-day delivery across Europe, same-day options within parts of the UK, and full chain-of-custody for compliant, traceable research.
Who are Research Donors?
Research Donors is an HTA-licensed clinic, based in London, dedicated to the collection and processing of human blood for research purposes. Research Donors is ISO 9001 2015 certified with Research Ethics (REC) approval as a Research Tissue bank, and participates in the UK NEQAS QA scheme.
Buffy coat sample validation data
|
|
|
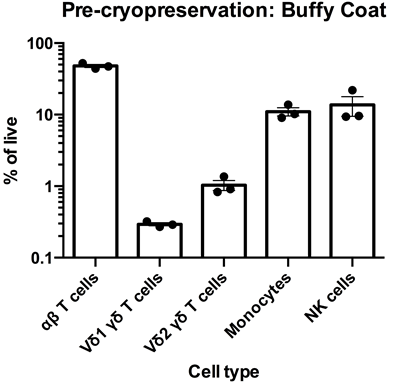
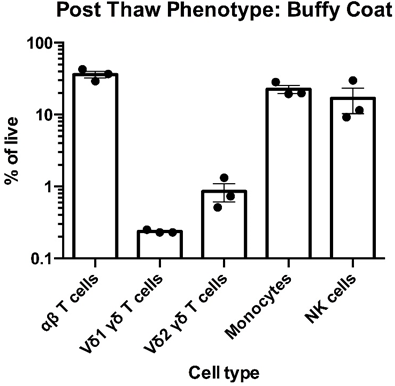
Left: Flow cytometry was used to assess the expression of CD3, TCR α/β TCR Vδ1, TCR Vδ2, CD14 and CD56 within PBMC freshly-isolated from buffy coats from three healthy donors. The data for αβT cells (CD3+TCR α/β +), Vδ1 yδT cells (CD3+TCR Vδ1+), Vδ2 yδT cells (CD3+TCR Vδ2+), monocytes (CD3-CD14+) and NK cells (CD3-CD56) is shown as mean percentage + SEM. Right: Flow cytometry was used to assess the expression of CD3, TCR α/β, TCR Vδ1, TCR Vδ2, CD14 and CD56 within PBMC immediately post cryorecovery in 10% DMSO from buffy coats from three healthy donors. The data for αβT cells (CD3+TCR α/β +), Vδ1 yδT cells (CD3+TCR Vδ1+), Vδ2 yδT cells (CD3+TCR Vδ2+), monocytes (CD3-CD14+) and NK cells (CD3-CD56) is shown as mean percentage + SEM.
Data courtesy of Jonathan Fisher et al, UCL Great Ormond Street Institute of Child Health, London, UK.
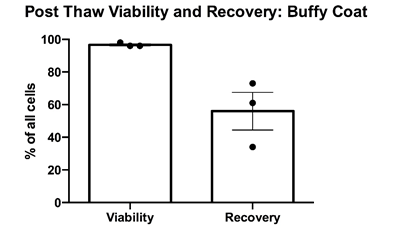
PBMCs were cryopreserved in 10% DMSO and post-thaw viability was determined via trypan blue exclusion. Cell counts pre- and post-cryopreservation were used to calculate the percentage recovery. Mean percentage + SEM and individual values are shown for PBMCs from buffy coats from three healthy donors. Cell recovery after cryopreservation was >50%.
Data courtesy of Jonathan Fisher et al, UCL Great Ormond Street Institute of Child Health, London, UK.
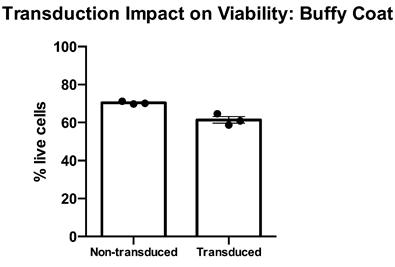
Thawed T cell expandibility, transducibility and the impact of transduction on T cell viability” – Cryopreserved PBMC from buffy coats from three healthy donors were thawed, rested overnight in complete media (RPMI1640 supplemented with L- Glutamine and 10% FCS) and stimulated with a translationally relevant T cell expansion cocktail; specifically, PBMC received a single does of TCR stimulus post-thaw and were cultured in complete media containing 100 IU/mL IL-2. The expansion was supplemented with fresh IL-2 containing media every 2-3 days. T cell viability was assessed using flow cytometry after 12 days of stimulation for cell viability. Day 12 overall cell viability was consistently >60% for all samples, with marginal impact post-transduction. Mean percentage + SEM and individual values are shown for n=3.
Data courtesy of Jonathan Fisher et al, UCL Great Ormond Street Institute of Child Health, London, UK.
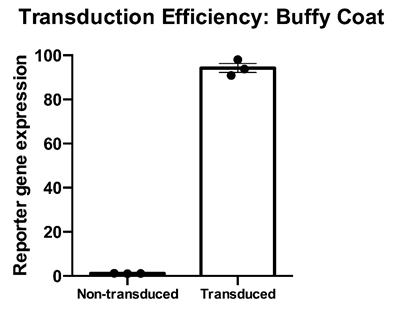
Thawed T cell transducibility” – Cryopreserved PBMC from three healthy donors were thawed, rested overnight in complete media (RPMI1640 supplemented with L- Glutamine and 10% FCS) and stimulated with a translationally relevant T cell expansion cocktail; specifically, PBMC received a single does of TCR stimulus post-thaw and were cultured in complete media containing 100 IU/mL IL-2. The expansion was supplemented with fresh IL-2 containing media every 2-3 days. T cell transducibility was assessed by viral transduction using a translationally-relevant lentiviral vector and protocol. T cell transduction efficiency was assessed on day 12 of expansion. T cells of buffy coats were higly and efficiently transducible with a lentiviralmultiplicity of infection of 4, with a mean efficiency of >90%. Data is shown for three donor buffy coat samples. Mean percentage + SEM and individual values are shown for n=3.
Data courtesy of Jonathan Fisher et al, UCL Great Ormond Street Institute of Child Health, London, UK.
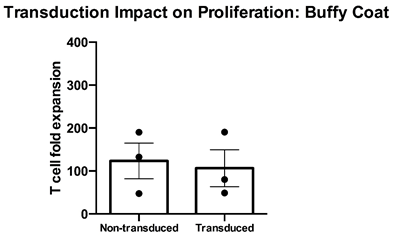
Thawed T cell expandability, transducibility and the impact of transduction on T cell viability” – Cryopreserved PBMC from three healthy donors were thawed, rested overnight in complete media (RPMI1640 supplemented with L- Glutamine and 10% FCS) and stimulated with a translationally relevant T cell expansion cocktail; specifically, PBMC received a single does of TCR stimulus post-thaw and were cultured in complete media containing 100 IU/mL IL-2. The expansion was supplemented with fresh IL-2 containing media every 2-3 days. T cell expansion was assessed using flow cytometry and counting beads after 12 days of stimulation for cell viability. While non-transduced buffy coat T cells expanded at a mean >120-fold, transduced T cells expanded to a similar extent at a mean 100-fold. Mean percentage + SEM and individual values are shown for n=3.
Data courtesy of Jonathan Fisher et al, UCL Great Ormond Street Institute of Child Health, London, UK.
These products are for use in Biomedical Research only. They are not to be used for any form of human treatment, or for any purpose that is prohibited by law, including any use for human reproductive cloning. It is your responsibility to ensure that you have necessary licensing and ethics approvals in place for the use and storage of any human tissue related material supplied by Cambridge Bioscience.