The patient-like Seraseq® reference standards from LGC Seracare provides consistent, defined reference material for Non-invasive prenatal testing (NIPT), one of the fastest growing test methods for screening pregnancies due to superior sensitivity compared to serum screening methods.
The NIPT technology is based on circulating maternal cell-free DNA enabling screening for fetal aneuploidies including trisomy 21, sex chromosome aneuploidies and some microdeletions. LGC Seracare's off-the-shelf and custom reference materials are developed using the innovative Seraseq innovative design process and support NIPT assay development, validation and quality control.
Benefits of using Seraseq NIPT reference standards
• Only patient-derived and patient-matched reference materials available on the market
• Manufactured in cGMP-compliant and ISO 13485-certified facilities
• NGS NIPT compatible, including SNP-based fetal fraction assays
• Products customisable regarding concentration, fetal fraction ration, format and volume
• Fast, easy to use and increase quality control consistency
• Eliminate the need to find, source and maintain remnant samples
Development of the maternal-fetal matched NIPT reference materials
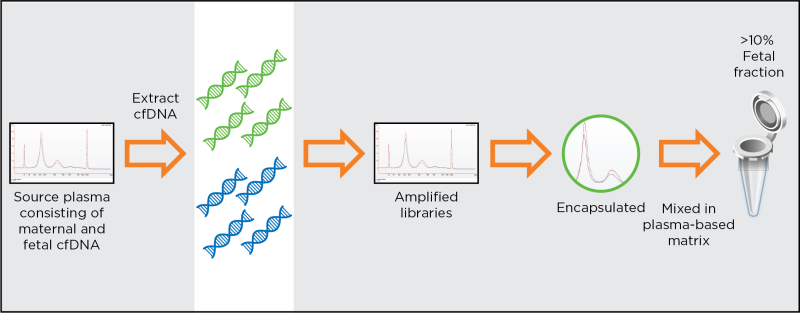
Unmatched patient-like materials to optmise, validate and standardise NIPT assays
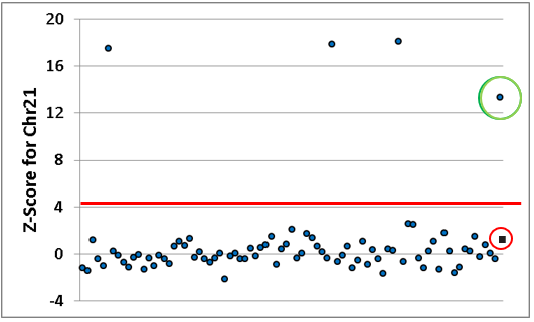
SeraSeq NIPT reference materials perform the same as patient's samples. The red line represents a cut-off of SD value. Circled in green and red are SeraSeq materials, trisomy 21 and euploid respectively
Material available for download
Reproductive health brochure
Seraseq NIPT reference materials product sheet

