The quantitative SARS-CoV-2 IgG ELISA from Wantai accurately quantifies levels of IgG antibodies to SARS-CoV-2 in human serum or plasma. Featuring an anti-SARS-CoV-2 immunoglobulin reference standard to allow the measurement of IgG levels, this quantitative ELISA enables researchers to identify individuals with an adaptive immune response to SARS-CoV-2 either as an indication of recent or prior infection, or as an aid in vaccination management decisions.
Benefits of using the SARS-CoV-2 IgG ELISA
• Levels of IgG antibodies to SARS-Cov-2 are accurately quantified
• Includes a reference standard calibrated against WHO approved standard (NIBSC code: 20/136)
• Test results issued in standardised units
• Aids in identifying individuals with an adaptive immune response to SARS-CoV-2
Clinical data
Sensitivity and specificity
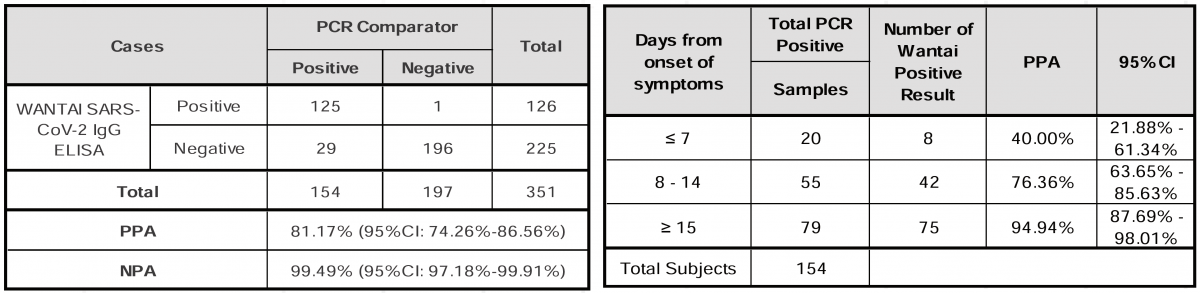
In a clinical study, of the total of 154 positive by PCR samples, 125 were positive on the WANTAI SARS-CoV-2 IgG ELISA (Quantitative), and of the 197 negative samples, 196 were negative. The kit demonstrated the Positive Percent Agreement (PPA) of 81.17% (125/154), the Negative Percent Agreement (NPA) of 99.49% (196/197). The kit demonstrated the PPA of 94.94% (75/79) for ≥ 15 days from onset of symptoms, as indicated in the tables below.
Performance data
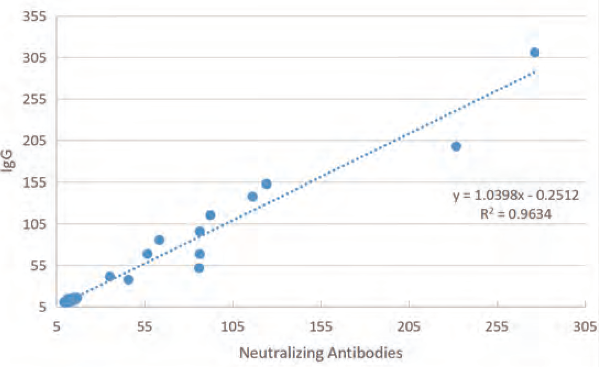
Retrospective analysis of the 75 positive samples (≥ 15 days from onset of symptoms) was conducted to measure the levels of IgG antibodies in the specimens. All specimens had IgG concentration of > 10.0 U/ml. Good correlation between the detected antibody levels with Wantai SARS-CoV-2 IgG (quantitative) ELISA and pseudovirus neutralisation test was established.
Cross reactivity
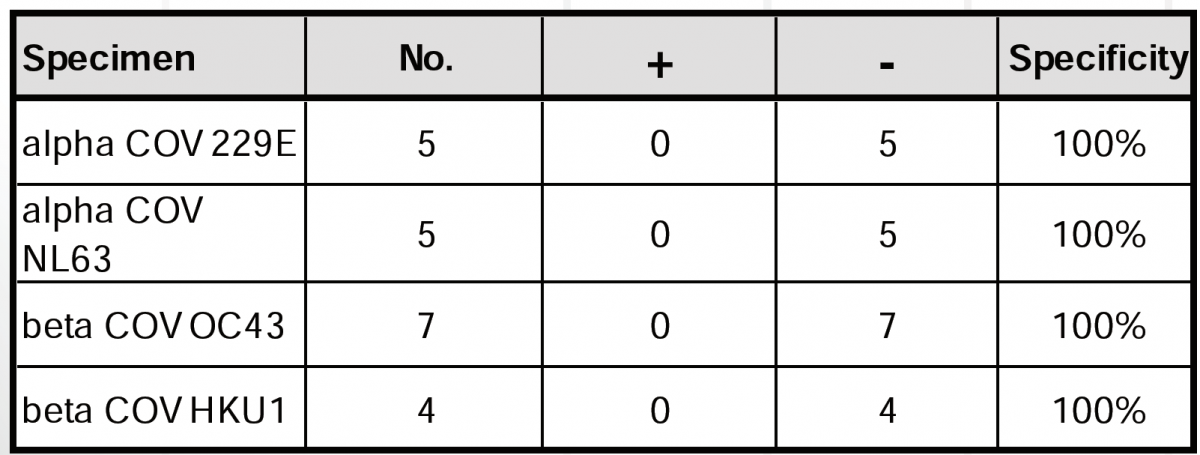
To evaluate the potential cross-reactivity of the quantitative SARS-CoV-2 IgG ELISA to antibodies to other viruses that may be present in the population, the following viruses were assessed.
Materials available for download
Quantitative SARS-CoV-2 IgG ELISA brochure
Quantitative SARS-CoV-2 IgG ELISA instructions for use
Products
Note: product availability depends on country - see product detail page.
| Details | Cat number & supplier | Size | Price |
| SARS-CoV-2 IgG ELISA (Quantitative) WS-1396 · Beijing Wantai Biological Pharmacy | WS-1396 Beijing Wantai Biological Pharmacy |
96 wells |
POA
96 wells
view
|

