ExoStd™ from Biovision are highly stable, highly purified exosome standards in a convenient lyophilised format. Extracted from human biological fluids (plasma, serum, urine and saliva pools from healthy donors) or from cell lines, ExoStd can be used as standardised positive controls for immunocapture performance evaluation, as control standards for multiple applications or as calibration standards for quantification of exosome-derived markers from biological samples.
Benefits of using ExoStd
• Highly purified using ultracentrifugation and microfiltration
• Lyophilised format maintains exosome composition
• Simple reconstitution in deionised water
• Lyophilised format stable for 36 months at 4°C
ExoStd purified and lyophilised exosomes

Purified and lyophilized exosomes are obtained from a variety of biological sources: cell culture supernatant, human plasma, serum, urine and saliva. Exosomes are purified following a combination of ultracentrifugation and microfiltration steps.
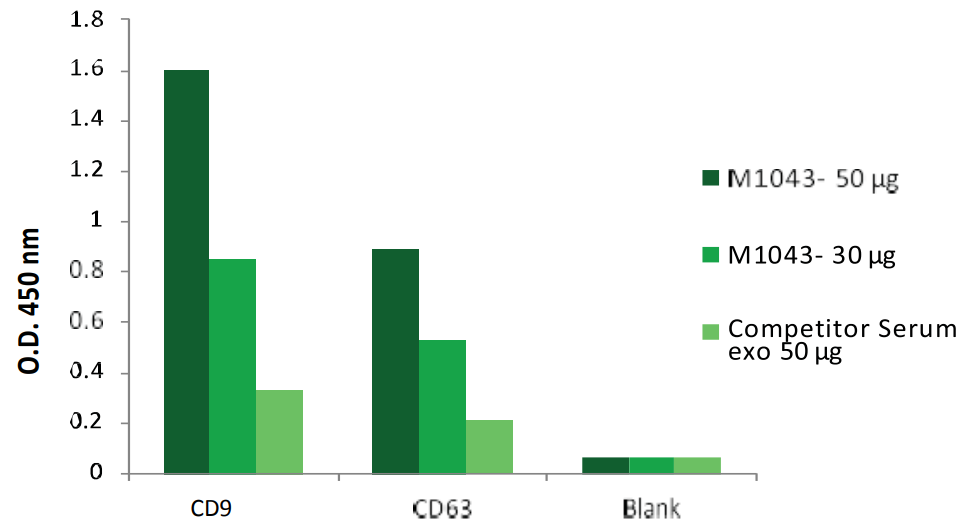
ELISA quantification of Biovision Exosome Standard vs Competitor’s standard
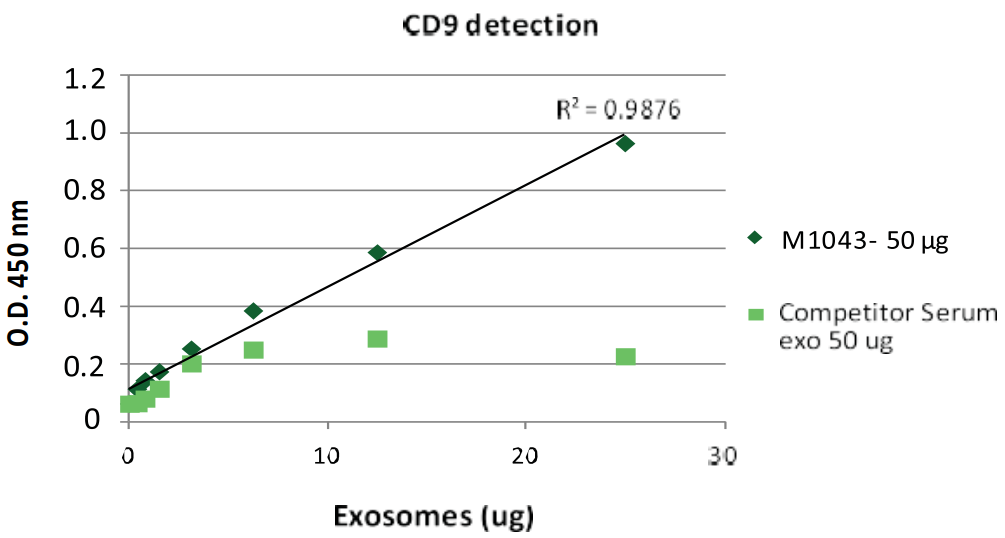
Standard curve for exosome quantification: Biovision Exosome Standard vs. Competitor’s Standard.
Applications
• Assay calibration
• Control (spike-in) for exosome quantification
• Protein marker analysis using different techniques
• Extraction and analysis of exosome nucleic acid
